The ability of the synchrotron to obtain time-resolved data allows the investigation of short-lived excited states, rapid structural changes in solid-state reactions and order/disorder phenomena that can be observed as they occur. Environmental cells can be used to probe materials under a range of non-ambient conditions. This allows characterisation of, for example, molecular packing, guests in a framework structure, the nature of intra- and intermolecular interactions, molecular conformation and static and dynamic disorder. Single crystal diffraction is the most widely used technique for obtaining full three-dimensional structural information of solid-state crystalline materials. Grazing Incidence X-ray Diffraction (GIXD).

The main diffraction techniques employed at Diamond are: If it’s crystallised first, then almost anything – from virus structures to ancient fossils – can be studied using diffraction. That’s why scientists use a technique called crystallography to freeze their samples into ice-like crystals. Some samples can be tricky to study using diffraction. It can be used to measure strains in materials under load, by monitoring changes in the spacing of atomic planes. This pattern can then be used to identify the atomic structure of the object itself.ĭiffraction patterns provide the atomic structure of molecules such as powders, small molecules or larger ordered molecules like protein crystals. This is because the light bounces off each atom inside the object, creating a unique arrangement of light and dark spots.

If you shine a bright light at an object, it produces a diffraction pattern as it leaves the sample. William Henry Bragg with the first spectrometer. The discoveries of von Laue and Bragg gave birth to two new sciences, X-ray crystallography and X-ray spectroscopy, and two Nobel Prizes: Max von Laue “for his discovery of the diffraction of X-rays by crystals” in 1914 and to Bragg and his father, Sir William Henry Bragg, “for their services in the analysis of crystal structure by means of X-rays” in 1915. Phenomenon to solve the first crystal structure and create a mathematical formula, Bragg’s Law, which showed how to work out the atomic structure of a sample based on the diffraction pattern it produced when exposed to X-rays. Within a year of this discovery, in 1912, William Henry Bragg and son Lawrence had exploited the The scatterers produced a regular array of spherical waves which gave the bright spots on the photographic plate. Physicists suspected that X-rays were a form of electromagnetic radiation, but had been unable to obtain solid evidence for their diffraction. Von Laue's experiment presented evidence for the wave nature of X-rays and the space lattice of crystals at the same time as the diffraction spots were caused by X-rays impinging on a regular array of scatterers, in this case, the repeating arrangement of atoms within the crystal. The discovery came 17 years after Wilhelm Conrad Röntgen had first demonstrated the existence of X-rays and their nature was still undetermined. He persuaded his colleagues Walter Friedrich and Paul Knipping – both of whom had more practical experience with X-rays than von Laue himself – to perform the experiment, the results of which showed diffraction spots surrounding the central spot of the primary beam. In 1912, Max von Laue and colleagues at the University of Munich, Germany came up with the idea to send a beam of X-rays through a copper sulfate crystal and record the results on photographic plates.
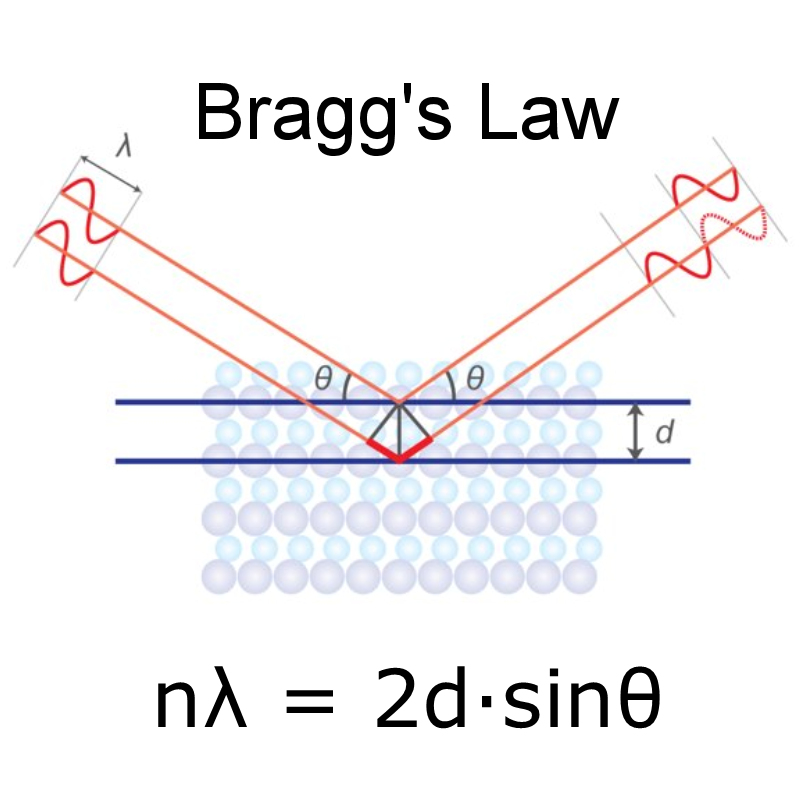
Wave diffraction was first observed in the 17th century, but it wasn’t until 1803, when Thomas Young performed an experiment to observe waves diffracting through two slits, that the phenomenon began to be more fully understood.
